Background: T-lineage acute lymphoblastic leukemia (T-ALL) and lymphoblastic lymphoma (T-LBL) arise from immature precursor T cells and are currently considered a single disease entity according to the World Health Organization Classification. Patients with T-ALL or T-LBL typically receive ALL-type intensive chemotherapy if applicable, and those with high-risk features or relapsed disease undergo hematopoietic cell transplantation (HCT). Although autologous HCT for T-ALL is uncommon, it remains a choice in the general practice of T-LBL, resulting in different donor selection strategies for these diseases. However, there is limited data on outcomes for these patients.
Methods: We analyzed nationwide registry data (Transplant Registry of Unified Management Program Japan, TRUMP) from 2000 to 2017 to evaluate outcomes after the first HCT and identify prognostic factors among p with T-ALL/T-LBL. Five patients who underwent autologous HCT for T-ALL were excluded from the analysis. Overall and disease-free survival (OS and DFS) were assessed using the Kaplan-Meier method with the log-rank test. Univariate and multivariate analyses were performed to identify the potential prognostic factors. The Cox proportional hazards method was used for the multivariate analysis to assess OS, whereas Gray's test and the Fine-Gray model were used to determine the cumulative incidence of relapse (CIR) considering competing risks such as relapse and non-relapse mortality (NRM).
Results: A total of 1345 patients (T-ALL, n=808; T-LBL, n=537) were included in the database. The median follow-up period of survivors was 4.5 (interquartile range, 2.0-7.7) years. The median age at HCT was 32 (range, 16-70) years. The OS and DFS at 4 years after HCT were comparable between T-ALL and T-LBL (OS 51.7% vs. 52.0%, p = 0.494; DFS 48.3% vs. 45.4%, p = 0.066). However, higher NRM and lower CIR were observed in T-ALL compared with T-LBL (4-year NRM 21.8% vs. 14.5%, p = 0.002; 4-year CIR 29.9% vs. 40.1%, p < 0.001). In T-LBL, patients who received allogeneic HCT (allogeneic LBL, n = 402) had significantly worse survival than those who received autologous HCT (autologous LBL, n = 135) (4-year OS 47.4% vs. 66.2%, p < 0.001; 4-year DFS 47.4% vs. 66.2%, p < 0.001), with significantly worse NRM and comparable CIR (4-year NRM 18.0 vs 3.8%, p < 0.001; 4-year CIR 39.6% vs. 41.6%, p = 0.875).
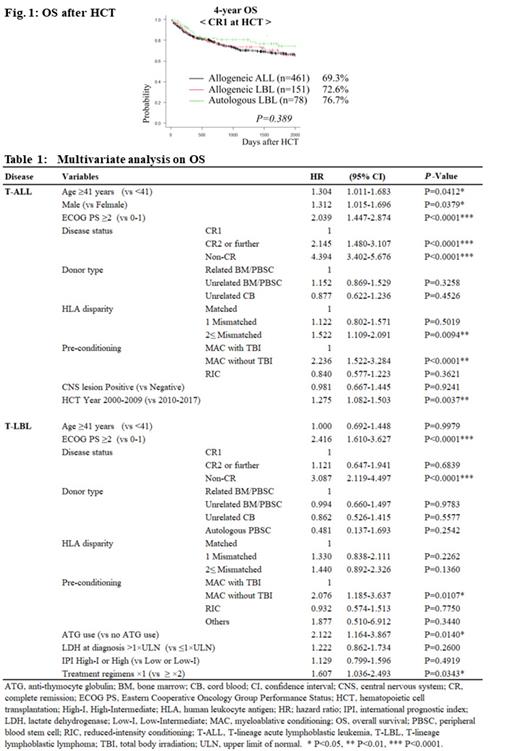
Compared with the allogeneic ALL or allogeneic LBL groups, the autologous LBL group had a significantly older age at HCT (the respective median age was 31 years [range, 16-70], 33 [16-86], and 49 [16-76]; p < 0.001). The allogeneic LBL group had a lower rate of first complete remission (CR1) and a higher rate of non-CR at SCT (CR1/non-CR: allogeneic ALL, 57.1%/30.4%; allogeneic LBL, 37.6%/47.0%; autologous LBL, 57.8%/31.1%; p < 0.001). Among patients in CR1 at HCT, OS and DFS were comparable among the allogeneic ALL (n = 461), allogeneic LBL (n = 151), and autologous LBL (n = 78) groups (Fig. 1, 4-year OS 69.3% vs. 72.6% vs. 76.7%, respectively, p = 0.389; 4-year DFS 64.9% vs. 65.5% vs. 67.1%, respectively, p = 0.830). However, the NRM was lower in the autologous LBL group (4-year NRM 16.6% vs. 8.3% vs. 0.0%, respectively, p < 0.001), and the CIR was lower in the allogeneic ALL group (4-year CIR 18.9% vs. 26.3% vs. 32.9%, respectively, p = 0.004). Among patients without CR at SCT, outcomes were comparable between allogeneic ALL (n = 246), allogeneic LBL (n = 189), and autologous LBL groups (n = 42) (4-year OS 18.4%, 24.6%, vs 36.9%, respectively, p = 0.347; 4-year DFS 17.4%, 21.9%, vs 23.6%, respectively, p = 0.818; 4-year NRM 29.8%, 28.0%, vs 12.7%, respectively, p = 0.107; 4-year CIR 52.8%, 50.2%, vs 63.7%, respectively, p = 0.347).
In T-ALL, multivariate analysis identified age ≥ 41 years, male sex, PS ≥ 2, disease status other than CR1, HLA disparity ≥ 2 loci, myeloablative conditioning (MAC) without total body irradiation (TBI), and HCT before 2010 as factors associated with poor OS (Table 1, Upper). In T-LBL, PS ≥ 2, non-CR at HCT, MAC without TBI, use of anti-thymocyte globulin, and multiple treatment regimens before HCT were significant factors for poor OS (Table 1, Lower). Donor type did not affect OS.
Conclusion: This largest-ever retrospective study shows comparable OS after HCT for T-ALL or T-LBL. Active disease, PS ≥ 2, and non-TBI MAC were potent poor prognostic factors. In T-LBL, autologous HCT can be a suitable option, which should be confirmed in further prospective studies.
Disclosures
Najima:Takeda Pharmaceutical Company Limited.: Speakers Bureau; Novartis Pharma K.K.: Speakers Bureau; Otsuka Pharmaceutical Co., Ltd.: Speakers Bureau; Nippon Shinyaku Co., Ltd.: Speakers Bureau; Sumitomo Pharma Co., Ltd.: Speakers Bureau; Astellas Pharma Inc.: Consultancy, Speakers Bureau; Kyowa Kirin Co., Ltd.: Speakers Bureau; Daiichi Sankyo Co. Ltd.: Consultancy, Speakers Bureau; AbbVie GK: Speakers Bureau; Amgen Inc.: Speakers Bureau; Janssen Pharmaceutical K.K.: Speakers Bureau; Bristol-Myers Squibb K.K.: Speakers Bureau; CSL Behring K.K.: Speakers Bureau; Chugai Pharmaceutical Co., Ltd.: Speakers Bureau. Suzuki:Takeda: Honoraria, Research Funding; Meiji Seika: Honoraria, Research Funding; Eisai: Honoraria, Research Funding; Shionogi: Honoraria, Research Funding; Sysmex: Research Funding; Taiho: Research Funding; Kyowa Kirin: Honoraria, Research Funding; Bristol Myers Squibb: Honoraria; Chugai Pharma: Honoraria, Research Funding; Sumitomo Dainippon Pharma: Honoraria; Novartis: Honoraria; Janssen: Honoraria; Abbvie: Honoraria; Otsuka: Honoraria, Research Funding; Nippon Shinyaku: Honoraria; AstraZeneca: Honoraria. Doki:Novartis Pharma K.K.: Honoraria; Janssen Pharmaceutical K.K.: Honoraria. Kurokawa:Otsuka Pharmaceutical Co., Ltd.: Honoraria, Research Funding; Eisai Co., Ltd.: Honoraria; Amgen Inc.: Honoraria; ONO PHARMACEUTICAL CO., LTD.: Honoraria; Kyowa Kirin Co., Ltd.: Honoraria, Research Funding; Sanofi K.K.: Honoraria; Sumitomo Pharma Food & Chemical Co., Ltd.: Honoraria, Research Funding; Nippon Shinyaku Co., Ltd.: Honoraria; Nippon Kayaku Co., Ltd.: Honoraria; DAIICHI SANKYO COMPANY, LIMITED: Honoraria, Research Funding; Takeda Pharmaceutical Company Limited.: Honoraria, Research Funding; Pfizer Japan Inc.: Honoraria; Bristol-Myers Squibb K.K.: Honoraria; MOCHIDA PHARMACEUTICAL CO.,LTD.: Honoraria; Janssen Pharmaceutical K.K.: Honoraria; Shionogi & Co., Ltd.: Research Funding; Teijin: Research Funding; Chugai Pharmaceutical Co., Ltd.: Honoraria, Research Funding; AbbVie GK: Honoraria, Research Funding; AstraZeneca K.K.: Research Funding; Astellas Pharma Inc.: Honoraria; Asahi Kasei Pharma Corporation: Honoraria, Research Funding; MSD K.K.: Honoraria. Kataoka:Eisai: Honoraria, Other: Scholarship; AbbVie: Honoraria; Novartis: Honoraria; Chugai Pharmaceutical: Honoraria, Other: Scholarship, Research Funding; AstraZeneca: Honoraria; Sumitomo Pharma: Honoraria, Other: Scholarship; Kyowa Kirin: Honoraria, Other: Scholarship; Janssen Pharmaceutical: Honoraria; Takeda Pharmaceutical: Honoraria, Other: Scholarship, Research Funding; SymBio Pharmaceuticals: Honoraria; Bristol Myers Squibb: Honoraria; Pfizer: Honoraria; Ono Pharmaceutical: Honoraria; Meiji Seika Pharma: Honoraria, Research Funding; Mundipharma: Honoraria; Sanofi: Honoraria; Sysmex: Honoraria; Nippon Shinyaku: Honoraria, Other: Scholarship; Daiichi Sankyo: Honoraria, Other: Scholarship; Alexion Pharmaceuticals: Honoraria; Incyte Corporation: Honoraria; Kyorin Pharmaceutical: Honoraria; Asahi Kasei Pharma: Other: Scholarship; Otsuka Pharmaceutical: Other: Scholarship, Research Funding; Shionogi: Other: Scholarship; Teijin Pharma: Other: Scholarship; Japan Blood Products Organization: Other: Scholarship; Mochida Pharmaceutical: Other: Scholarship; JCR Pharmaceuticals: Other: Scholarship; Nippon Kayaku: Other: Scholarship; Chordia Therapeutics: Research Funding. Kanda:Wakunaga Pharmaceutical: Speakers Bureau; FUJIFILM Wako Pure Chemical: Speakers Bureau; Nippon Shinyaku: Speakers Bureau; Towa Pharma: Speakers Bureau; AbbVie: Research Funding, Speakers Bureau; CSL Behring: Speakers Bureau; Japan Blood Products Organization: Research Funding, Speakers Bureau; Otsuka Pharmaceutical: Research Funding, Speakers Bureau; AstraZeneca: Speakers Bureau; Human Life CORD: Speakers Bureau; Sumitomo Pharma: Research Funding, Speakers Bureau; Amgen: Speakers Bureau; Takeda Pharmaceutical: Research Funding, Speakers Bureau; Meiji Seika Pharma: Speakers Bureau; Asahi Kasei Pharma: Research Funding, Speakers Bureau; Daiichi Sankyo: Research Funding, Speakers Bureau; Saitama Hokeni Kyokai: Speakers Bureau; MSD: Speakers Bureau; Kyowa Kirin: Research Funding, Speakers Bureau; Janssen Pharmaceutical: Speakers Bureau; Sanofi: Speakers Bureau; Pfizer: Speakers Bureau; Chugai Pharmaceutical: Research Funding, Speakers Bureau; Novartis: Speakers Bureau; Bristol Myers Squibb: Speakers Bureau; Eisai: Research Funding, Speakers Bureau; Precision: Speakers Bureau; Alexion Pharma: Speakers Bureau; Shionogi Pharma: Research Funding; Taiho Pharmaceutical: Research Funding; Nippon Kayaku: Research Funding; JCR Pharmaceuticals: Research Funding. Sawa:Janssen: Honoraria; Sanofi: Honoraria. Ichinohe:Kyowa Kirin Co.: Research Funding; Chugai Pharmaceutical Co.: Research Funding; Takeda Pharmaceutical Co.: Research Funding; Ono Pharmaceutical Co.: Research Funding; Nippon Shinyaku Co.: Research Funding; Repertoire Genesis Inc.: Research Funding. Atsuta:Meiji Seika Pharma Co, Ltd.: Honoraria; Novartis Pharma KK: Speakers Bureau; CHUGAI PHARMACEUTICAL CO., LTD.: Speakers Bureau; JCR Pharmaceuticals Co., Ltd.: Consultancy; Otsuka Pharmaceutical Co., Ltd: Speakers Bureau.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal